Hepatology, December 1999, p. 1529-1530, Vol. 30, No. 6
Correspondence
Pain Experienced During Percutaneous Liver Biopsy
To the Editor:
Percutaneous liver biopsy for the diagnosis of liver disease is a well-established, widely and routinely used procedure with low morbidity.1 However, it is invasive and often considered painful by patients. We conducted a pilot study aimed at grading the intensity of pain experienced by patients during liver biopsy to determine whether or not prophylactic analgesia might be needed.
Thirty consecutive adult patients (19 men, 11 women; mean age 42 ± 11 years) undergoing a first percutaneous liver biopsy (for chronic Hepatitis C in 22; alcohol-induced liver disease in 5; miscellaneous in 3) were included after written informed consent was given. Exclusion criteria were (1) abdominal ultrasound revealing a contraindication such as intrahepatic bile duct dilatation, focal liver lesion, hydatid disease, or massive ascites; (2) clotting disorders such as prothrombin less than 50% of normal or cephalin kaolin time exceeding 1.5 times the normal control time, or platelet count below 105/mm3, or Ivy’s bleeding time exceeding 4 minutes; (3) ongoing analgesic treatment; (4) comprehension difficulty. Before starting the procedure patients were taught in a standard way by the same investigator how to use a visual analogue scale (VAS) scoring from 0 to 100 mm to grade the intensity of pain experienced during liver biopsy. On that scale, the left end point, 0, was defined as no pain and the right end point, 100, as the worst pain the patient could imagine. There were no further marks on the line. The intensity of pain was indicated by the distance in millimeters from the left end point. Liver biopsy was performed at bedside according to the Menghini technique2 by the same senior operator after adequate local anesthesia with lidocaine 1% (10 mL). Patients were asked to grade on the VAS the intensity of pain experienced during liver biopsy, at the end of procedure (D0), and the next day at discharge (D1) to validate the reproducibility of results.
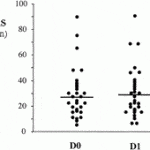
All liver specimens could be obtained at first passage. Mean VAS pain scores were 28 ± 3 mm (range, 5-91) at D0 and 30 ± 4 mm (range, 6-95) at D1, respectively, (r = .78; P < .0001) (Fig.1). Six out of 30 patients (20%) experienced severe pain (i.e. VAS >40 mm). All were men, with chronic Hepatitis C in 5 cases.
| Fig. 1. Pain scores measured by the VAS at the end of the procedure (D0) and the next day (D1). Individual data; mean values are indicated by the line. |
The intensity of pain experienced during biopsy has not been specifically studied in the literature probably because pain is usually short in duration and deemed as minor by most hepatologists. However, we have observed using the VAS, the validated and recommended method to grade pain,3 that the mean pain score at D0 was close to 30 mm with a maximum range of 91. Similar results were obtained at D1. Such a pain score, similar to that of other minor procedures, such as venous cannulation, is usually considered mild to moderate and requires analgesia.4 Furthermore, 20% of the patients experienced severe pain during liver biopsy. Most of these patients had chronic Hepatitis C, the main indication for liver biopsy in our series like in many other centers. Patients with chronic Hepatitis C are usually young and asymptomatic and thus consider liver biopsy a major ordeal. Moreover, during the course of chronic Hepatitis C, repeated liver biopsies may be necessary for management of patients.5 Those who experienced severe pain and were not provided adequate analgesia may thus be lost to follow-up after the first biopsy. Over the past decade, the importance of liver biopsy for diagnosis, therapeutic decisions, and for monitoring has considerably grown with liver transplantation and the use of interferon alfa for chronic Hepatitis B and C. It is likely that the number of liver biopsies will continue to increase in the coming years, with over 1 million people infected by Hepatitis C virus worldwide,6 with the promising results of the combination of ribavirin and interferon alfa for treatment of patients with chronic Hepatitis C,7,8 and with the need to repeat liver biopsy during follow-up of patients with chronic liver disease.
In conclusion, the results of this pilot study suggest that, although deemed as minor, pain experienced during percutaneous liver biopsy should be taken into consideration and that patients should be provided adequate prophylactic analgesia.
Acknowledgment
The authors are grateful to Professor Dan Benhamou for his helpful comments.
Laurent Castéra, M.D.
Isabelle Nègre, M.D.
Kamran Samii, M.D.
Catherine Buffet, M.D.
Department of Hepatology and Gastroenterology,
Department of Anesthesia and General Intensive CareHôpital BicêtreUniversité Paris-Sud,Le Kremlin-Bicêtre, France
REFERENCES
Copyright © 1999 by the American Association for the Study of Liver Diseases.