What Is Liver Dialysis and Who Can Benefit From It?
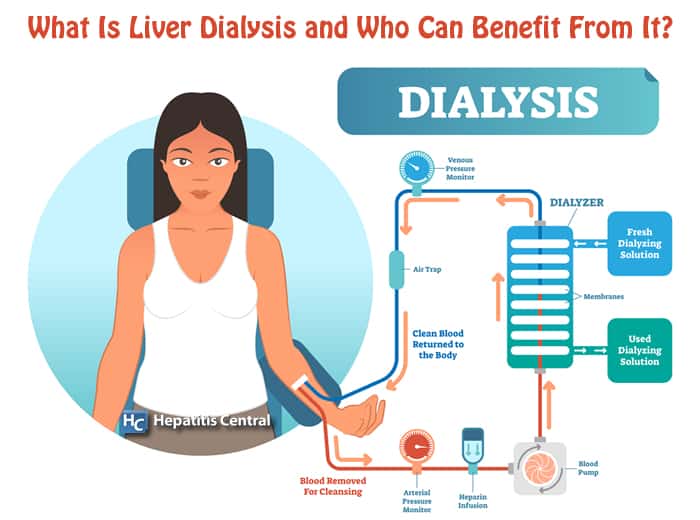

What Is Liver Dialysis?
Based on the same principles as kidney dialysis, liver dialysis is a detoxification treatment for acute liver failure. Liver dialysis has existed since the early 2000s. However, the applicability and affectivity of liver dialysis has been subject to mixed reviews.
While kidney dialysis is used for kidney failure and primarily removes water-soluble toxins, it does not, however, remove toxins bound to albumin that accumulate in liver failure. Thus, liver dialysis requires a different model.
Similar to a bio-artificial liver device, liver dialysis is a form of artificial extracorporeal (outside of the body) liver support.
Although the liver performs many life-sustaining functions, a critical issue of liver failure is the accumulation of toxins not cleared by the failing liver.
Based on this, the removal of the following lipophilic, albumin-bound substances is assumed to benefit a person in liver failure:
- Bilirubin
- Bile acids
- Metabolites of aromatic amino acids
- Medium-chain fatty acids
- Cytokines
Liver Dialysis Devices
There are several devices designed to artificially detoxify the liver, including the Molecular Adsorbent Recirculating System (MARS), Single Pass Albumin Dialysis (SPAD) and the Prometheus system.
- Molecular Adsorbents Recirculation System (MARS) – MARS was developed by Teraklin AG of Germany about ten years ago, and is the best-known extracorporeal liver dialysis system. MARS is a blood detoxification system with two separate dialysis circuits that is based on albumin dialysis and is able to remove albumin-bound and water-soluble substances selectively. As of 2005, MARS has 510 (k) approval from the Food and Drug Administration (FDA) for drug overdoses and poisoning. While MARS is the widely accepted albumin dialysis technique, it has limited use in developing countries because of its technical difficulties and high cost.
- Single Pass Albumin Dialysis (SPAD) – SPAD is a simple method of albumin dialysis using standard renal replacement therapy machines without the additional perfusion pump system that MARS has.
- Prometheus – The Prometheus system by Fresenius Medical Care is a newer device in which albumin-bound substances are directly removed from the blood by a special adsorber. In a simultaneous step, high-flux hemodialysis is performed. Hard data for the use of Prometheus in acute liver failure is still lacking.
6 More Important Facts About Liver Dialysis
Liver dialysis treatment is in its infancy, with ongoing trials underway worldwide.
- A relatively small number of hospitals in the U.S. offer liver dialysis. Although not exclusive, locations include Jefferson Hospital in Philadelphia, PA, Sharp Coronado Hospital in San Diego CA, Cincinnati Children’s Hospital Medical Center in Cincinnati, OH, University of Maryland Medical Center in MD, Emory University Hospital, Atlanta, GA, and Texas Children’s Hospital in Houston, TX.
- Liver dialysis’s intended use is to buy time for people with acute liver failure awaiting liver transplantation or recovery of liver function.
- MARS is approved by the FDA for use in acute liver failure caused by drug or toxins.
- Unlike hemodialysis for kidney failure, liver dialysis cannot support a patient for an extended period of time (months to years).
- Liver dialysis can last up to six hours, but patients may only need one or two sessions.
- Although MARS can reduce the effects of brain swelling related to liver failure, it is not FDA-approved as a bridge to liver transplant.
Liver Dialysis Trial Results
Because liver dialysis is relatively new, there are no crystal clear recommendations for its use and there is a murky understanding of the risks involved.
As reported in the journal, Advances in Chronic Kidney Disease, a 2015 systematic review by Boston researchers examined the efficacy and safety of MARS, Prometheus and SPAD for liver failure.
By meta-analysis on ten trials:
- Albumin dialysis achieved a net decrease in serum total bilirubin level relative to standard medical therapy, but no net decrease in serum ammonia or bile acids.
- Albumin dialysis achieved an improvement in hepatic encephalopathy relative to standard medical therapy, but had no effect on survival.
- Because of inconsistency in the reporting of adverse events, the safety evaluation was inconclusive.
- Albumin dialysis as a supportive treatment for liver failure is successful at removing albumin-bound molecules, such as bilirubin and at improving hepatic encephalopathy.
As published in a January 2016 issue of the journal Critical Care, German researchers compared MARS with SPAD liver dialysis methods. According to the authors, both procedures were safe for temporary extracorporeal liver support.
While in clinical practice routinely assessed plasma bilirubin levels were reduced by both systems, only MARS affected other para-clinical parameters (i.e., serum bile acids, albumin-binding capacity, and creatinine and urea levels).
Caution should be taken with regard to metabolic derangements and electrolyte disturbances, particularly in SPAD using regional citrate anti-coagulation.
Who Gets MARS Therapy?
One of the challenges of this seemingly idealistic treatment is determining who will benefit from MARS therapy.
According to Thomas M. Scalea, MD, the Honorable Francis X. Kelly Distinguished Professor of Trauma Surgery and director of the Program in Trauma at the University of Maryland School of Medicine, and Physician-in-Chief, R Adams Cowley Shock Trauma Center, “MARS therapy seems to improve the biochemical parameters. That seems pretty clear. In people with significant biochemical abnormalities that have a potentially reversible cause of their liver failure, or who are considered good candidates for transplant, it seems like a good idea. On the other hand, MARS is not very helpful for people with multiple organ failure.”
According to the director of the University of Maryland MARS program, Deborah M. Stein, MD, MPH, the R Adams Cowley Professor of Trauma Surgery at the University of Maryland School of Medicine and chief of trauma and director, Neurotrauma Critical Care at STC, the doctors look at the following factors to determine eligibility:
- overall patient survivability
- degree and cause of liver dysfunction
- the degree of coexisting organ failure
In general, it appears that the use of MARS is allocated to cases of acute liver failure in young and previously healthy people. As such, it is typically not considered for those enduring a chronic liver disease battle (such as chronic Hepatitis C).
Conclusion
Skeptics say that, while liver dialysis treatment may help, there’s not enough data to show whether it gives patients a better outcome, a shorter hospital stay, or a longer life than traditional drug therapies.
While the MARS system may help keep a person alive while they await a liver transplant, dialysis machines only filter the blood – they can’t take over other liver functions.
Lest we forget, the liver performs hundreds of life sustaining functions. While liver dialysis may be valuable to a small subset of individuals in acute liver failure, the majority of those with a failing liver are stuck waiting for a coveted liver transplant.
https://ccforum.biomedcentral.com/articles/10.1186/cc2895, Equipment review: The molecular adsorbents recirculating system (MARS®), M. Boyle, et al, Retrieved October 28, 2018, Critical Care, 2004.
https://ccforum.biomedcentral.com/articles/10.1186/s13054-015-1159-3, Molecular adsorbent recirculating system and single-pass albumin dialysis in liver failure – a prospective, randomised crossover study, C. Sponholz, et al, Retrieved October 29, 2018, Critical Care, January 2016.
https://craigcameron.us/what-is/458-what-is-liver-dialysis/, What is Liver Dialysis?, Craig, Retrieved October 28, 2018, Living with Liver Disease, 2015.
https://hospitals.jefferson.edu/tests-and-treatments/liver-dialysis.html, Liver Dialysis, Retrieved October 27, 2018, Thomas Jefferson University Hospitals, 2018.
https://www.americannursetoday.com/mars-the-new-frontier-in-treating-acute-liver-failure/, MARS®: The new frontier in treating acute liver failure, M.D. Still, Retrieved October 28, 2018, American Nurse Today, March 2014.
https://www.cincinnatichildrens.org/health/m/mars-therapy-liver-dialysis, MARS Therapy (Liver Dialysis), Retrieved October 28, 2018, Cincinnati Children’s Hospital Medical Center, 2018.
https://www.eurekalert.org/pub_releases/2017-07/eaft-fpt072117.php?fbclid=IwAR3iAU9ELw4ri10HEMW5kBkwWJuGGc8cLGbKdwnhayJ9vOS6MS6MvNvDH7U, First patient treated in trial of a novel liver dialysis device, DIALIVE, European Association for the Study of the Liver, Retrieved October 27, 2018, American Association for the Advancement of Science, 2017.
https://www.kidney.org/atoz/content/hemodialysis, Hemodialysis, Retrieved October 28, 2018, National Kidney Foundation, 2018.
https://www.ncbi.nlm.nih.gov/pubmed/26311600, Albumin Dialysis for Liver Failure: A Systematic Review, Tsipotis, E, et al, Retrieved October 28, 2018, Advances in Chronic Kidney Disease, September 2015.
https://www.sciencedirect.com/topics/medicine-and-dentistry/liver-dialysis, Liver Dialysis, Retrieved October 28, 2018, Elsevier B.V., 2018.
https://www.sharp.com/hospitals/coronado/departments/mars-treatment.cfm, MARS Liver Dialysis Treatment, Retrieved October 28, 2018, Sharp HealthCare, 2018.
https://www.umms.org/ummc/news/2017/mars-study, New Use of Blood Cleaning Device Saves High-Risk Patients With Liver Failure, Retrieved October 29, 2018, University of Maryland Medical Center, 2018.
https://www.usatoday.com/story/news/nation/2014/05/28/liver-dialysis-life-saving-treatment/9674977/, Liver Dialysis Proves Another Life-Saving Procedure, Mark Wert, Retrieved October 29, 2018, USA Today, 2018.





