Determinants of Outcome of Compensated Hepatitis C Virus-Related Cirrhosis
HEPATOLOGY, May 1998, p. 1435-1440, Vol. 27, No. 5
Original Articles
Lawrence Serfaty1, Hugues Aumaître1, Olivier Chazouillères1, Anne-Marie Bonnand2, Olivier Rosmorduc1, Renée E. Poupon2, and Raoul Poupon1
From the 1 Service d’Hépato-gastroentérologie, Hôpital St-Antoine, Assistance Publique-Hôpitaux de Paris, Paris, and 2 INSERM Unit 21, Villejuif, France
ABSTRACT
The aim of this study was to assess the incidence of decompensation (ascites, jaundice, variceal bleeding, and encephalopathy), hepatocellular carcinoma (HCC) and death or liver transplantation in patients with compensated Hepatitis C virus (HCV)-related cirrhosis, taking into account the viral genotype and interferon (IFN) therapy. Between 1989 and 1994, 668 patients with no clinical evidence of decompensation were referred to our department for liver biopsy because of positivity for anti-HCV antibodies and elevated aminotransferase activity; 103 of these patients had cirrhosis. The median follow-up was 40 months. Fifty-nine patients were treated with IFN for a mean duration of 11 ± 6 months; 3 (5%) had a prolonged biochemical and virological response. Baseline characteristics of IFN-treated and untreated patients were not significantly different. HCV genotypes (InnoLiPa) were predominantly 1b (48%) and 3a (20%). During follow-up, complications of cirrhosis occurred in 26 patients, HCC in 11 patients, and decompensation not related to HCC in 19 patients. Sixteen patients died, 94% of liver disease. Three patients were transplanted for liver failure. The 4-year risk of HCC was 11.5% (annual incidence 3.3%) and that of decompensation was 20%. Survival probability was 96% and 84% at 2 and 4 years, respectively. In multivariate analysis, the absence of IFN therapy was the only independent factor predictive both for HCC and decompensation. A low albumin level at entry and the absence of IFN therapy were the two independent factors predictive of death or liver transplantation. Probability of survival at 2 and 4 years was significantly different between IFN-treated and untreated patients (respectively 97% and 92% vs 95% and 63%, P < .0001). In conclusion, in patients with compensated HCV-related cirrhosis: 1) complications of cirrhosis are frequent, whatever the viral genotype; and 2) the severity of cirrhosis and the absence of IFN therapy are independently predictive of bad outcome. (HEPATOLOGY 1998;27:1435-1440.)
INTRODUCTION
Hepatitis C virus (HCV) infection is one of the main causes of death related to cirrhosis and liver transplantation in France.1 The incidence of hepatocellular carcinoma (HCC) and decompensation is not precisely known, particularly in Europe and the United States. Indeed, study populations are often heterogeneous in terms of the severity of liver disease, as well as interferon (IFN) treatment.2-11 The long-term mortality rate among patients with chronic Hepatitis C is controversial. 2,8 Prognostic factors for complications of HCV-related cirrhosis are not clearly defined, particularly the role of the HCV genotype.12-15 Recent studies have suggested that IFN therapy reduces the risk of HCC in HCV-related cirrhosis. 16,17 The aim of this study was to assess in a cohort of patients with compensated HCV-related cirrhosis the following: 1) the incidence of decompensation (ascites, jaundice, variceal bleeding, and encephalopathy), HCC, and death or liver transplantation; and 2) factors predictive of complications, including the HCV genotype and IFN therapy.
PATIENTS AND METHODS
Patients
Study Population. Between December 1989 and December 1994, 668 patients with no clinical evidence of decompensation were referred to our department for liver biopsy because of anti-HCV seropositivity (enzyme-linked immunosorbent assay and radio immunoblot assay) and elevated alanine aminotransferase (ALT) activity. All patients meeting the following criteria were included in the study: 1) histological diagnosis of cirrhosis defined by nodular regeneration; 2) no history or clinical sign at entry into the study of complications of cirrhosis, ie, ascites, jaundice (serum bilirubin > 3 mg/dL (51 µmol/L)), encephalopathy, or variceal bleeding; 3) no evidence of HCC at entry into the study on the basis of ultrasonography and ![]() -fetoprotein level < 500 µg/L; 4) absence of serum Hepatitis B surface antigen; 5) absence of co-existing liver disease such as hemochromatosis, Wilson’s disease, or biliary cirrhosis; 6) absence of excessive alcohol consumption (> 80 g per day); and 7) absence of human immunodeficiency virus antibodies.
-fetoprotein level < 500 µg/L; 4) absence of serum Hepatitis B surface antigen; 5) absence of co-existing liver disease such as hemochromatosis, Wilson’s disease, or biliary cirrhosis; 6) absence of excessive alcohol consumption (> 80 g per day); and 7) absence of human immunodeficiency virus antibodies.
One hundred and three patients fulfilling these criteria were included in the study. Fifty nine of these patients (57%) were treated with recombinant IFN-![]() 2a or IFN-
2a or IFN-![]() 2b (3 MU × 3/week) for a mean of 11 months (range, 6-33 months). A response to treatment was defined by persistent normalization of ALT activity and absence of serum HCV RNA one year after the end of treatment.
2b (3 MU × 3/week) for a mean of 11 months (range, 6-33 months). A response to treatment was defined by persistent normalization of ALT activity and absence of serum HCV RNA one year after the end of treatment.
Follow-Up. Entry into the study was defined as the time of diagnosis of compensated HCV-related cirrhosis, provided that patients fulfilled the inclusion criteria. The follow-up period ended at the date of death or liver transplantation or was censored at the last recorded visit before January 1996. All patients were examined every 6 months or more frequently, if indicated. Routine evaluation included a physical examination, standard biochemical liver tests, ![]() -fetoprotein assay, and liver ultrasound examination. The following events were defined as severe clinical complications: death, liver transplantation, onset of ascites, jaundice (serum bilirubin > 3 mg/dL (51 µmol/L), hepatic encephalopathy, bleeding from esophageal varices (hematemesis and/or melena), and HCC. Decompensation was considered to be related to HCC if decompensation and HCC were diagnosed at the same time. The diagnosis of HCC, based on increasing levels of
-fetoprotein assay, and liver ultrasound examination. The following events were defined as severe clinical complications: death, liver transplantation, onset of ascites, jaundice (serum bilirubin > 3 mg/dL (51 µmol/L), hepatic encephalopathy, bleeding from esophageal varices (hematemesis and/or melena), and HCC. Decompensation was considered to be related to HCC if decompensation and HCC were diagnosed at the same time. The diagnosis of HCC, based on increasing levels of ![]() -fetoprotein, and ultrasound findings, was always confirmed histologically.
-fetoprotein, and ultrasound findings, was always confirmed histologically.
Methods
Laboratory Tests. Viral parameters were studied on sera stored at -80°C, by means of the Amplicor* HCV RNA qualitative test (Roche Diagnostic), a standardized reverse transcription-polymerase chain reaction (PCR) assay, according to the manufacturer’s instructions.
To determine the HCV genotype, PCR products from the Amplicor* assay were directly analyzed by using a reverse hybridization assay (InnoLipa, HCV, Innogenetics), according to the manufacturer’s instructions.18 In case of PCR-negativity, the serotype was determined by using an enzyme-linked immunosorbent assay technique (Murex’ HCV serotyping 1-6 assay). Genotypes were classified according to Simmonds.19 Data for patients infected by genotypes other than 1b were pooled and compared with those for patients infected by genotype 1b strains.
Prognostic Factors. The following clinical, biochemical, virological, and histological variables measured at the time of liver biopsy were evaluated as potential prognostic factors for severe clinical complications: age at diagnosis, sex, history of transfusion or iv drug use, duration of HCV infection in patients with known risk factors (calculated from the date of transfusion or the beginning of iv drug use), hepatomegaly and splenomegaly, body/mass index, aspartate aminotransferase, ALT, gammaglutamyl transpeptidase, alkaline phosphatase, albumin, bilirubin, prothrombin time, platelets count, Child-Pugh score, -fetoprotein, anti-Hepatitis B core antibody, infection with genotype 1b, and the Knodell score.20 The prognostic value of IFN therapy for severe clinical complications was also analyzed.
Statistical Analysis. Descriptive statistics are provided as means ± SD. Fisher’s Exact test or the Mann-Whitney test was used for statistical comparisons. In the overall group, time to occurrence of HCC, decompensation unrelated to HCC, and death or liver transplantation were estimated using the Kaplan-Meier method.
For each continuous parameter, a cutoff was defined to divide the patients into 2 groups with approximately equal numbers of events. Probabilities of HCC, decompensation unrelated to HCC, and survival were estimated in each group using the Kaplan-Meier method.21 The univariate associations between potentially predictive variables and the 3 endpoints were tested with the log-rank test. The independent predictive value of each parameter was assessed with a stepwise multivariate Cox model.22 In both univariate and multivariate analyses, the relative risks were calculated by using a Cox regression model. A P value of less than .05 was considered to denote a significant difference. The univariate and multivariate analyses were performed using the BMDP package.
RESULTS
Patient’s Characteristics at the Time of Liver Biopsy
One hundred and three patients with compensated HCV-related cirrhosis fulfilled the inclusion criteria. Fifty nine of these patients were treated with recombinant IFN-![]() , 44 were untreated. Baseline characteristics of IFN-treated and untreated patients were not significantly different (table 1). Serum HCV RNA was detected by RT-PCR in 97 patients. Among these 97 patients, the HCV genotypes were as follows: 1a in 16 (16%); 1b in 49 (51%); 2a in 6 (6%); 3a in 20 (21%); mixed in 1 (1%); and undetermined in 5 (5%). Among the 6 PCR-negative patients, the serotype was 1 in three cases, 2 in two, and undetermined in one. Forty nine (48%) of the 103 patients were considered to be infected by the genotype 1b strain.
, 44 were untreated. Baseline characteristics of IFN-treated and untreated patients were not significantly different (table 1). Serum HCV RNA was detected by RT-PCR in 97 patients. Among these 97 patients, the HCV genotypes were as follows: 1a in 16 (16%); 1b in 49 (51%); 2a in 6 (6%); 3a in 20 (21%); mixed in 1 (1%); and undetermined in 5 (5%). Among the 6 PCR-negative patients, the serotype was 1 in three cases, 2 in two, and undetermined in one. Forty nine (48%) of the 103 patients were considered to be infected by the genotype 1b strain.
| table 1. Baseline Characteristics of Interferon-Treated Patients and Untreated Patients |
Follow-Up of Patients
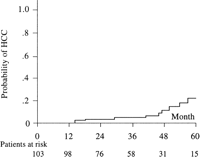
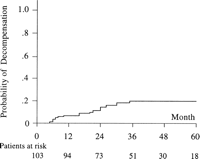
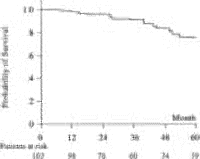
The median follow-up was 40 months (range, 6-72 months). Three patients (3%) were lost to follow up. Among the 59 patients treated with recombinant IFN-![]() 2, 6 (10%) had a sustained normalization of ALT activity and 3 (5%) had a sustained virological response. During follow up, 26 patients (25%) developed complications of cirrhosis, with HCC in 11 (11%) and decompensation unrelated to HCC in 19 (4 patients decompensated before HCC occurrence). The cumulative probabilities of developing HCC were 3% and 11.5% at 2 and 4 years, respectively (Fig. 1); the annual incidence of HCC was 3.3%. The cumulative probabilities of developing decompensation unrelated to HCC were 15% and 20% at 2 and 4 years, respectively (Fig. 2). Sixteen patients (16%) died during the follow up. The causes of death were liver failure in 5, HCC in 9, variceal bleeding in 1, and nonhepatic in 1. Three patients were transplanted for liver failure. The cumulative probabilities of survival were 96% and 84% at 2 and 4 years, respectively (Fig. 3). The annual incidence of death or transplantation was 5.5%. In the 19 patients who died or were transplanted, the time between the first complication of cirrhosis and death or liver transplantation ranged from 2 to 37 months.
2, 6 (10%) had a sustained normalization of ALT activity and 3 (5%) had a sustained virological response. During follow up, 26 patients (25%) developed complications of cirrhosis, with HCC in 11 (11%) and decompensation unrelated to HCC in 19 (4 patients decompensated before HCC occurrence). The cumulative probabilities of developing HCC were 3% and 11.5% at 2 and 4 years, respectively (Fig. 1); the annual incidence of HCC was 3.3%. The cumulative probabilities of developing decompensation unrelated to HCC were 15% and 20% at 2 and 4 years, respectively (Fig. 2). Sixteen patients (16%) died during the follow up. The causes of death were liver failure in 5, HCC in 9, variceal bleeding in 1, and nonhepatic in 1. Three patients were transplanted for liver failure. The cumulative probabilities of survival were 96% and 84% at 2 and 4 years, respectively (Fig. 3). The annual incidence of death or transplantation was 5.5%. In the 19 patients who died or were transplanted, the time between the first complication of cirrhosis and death or liver transplantation ranged from 2 to 37 months.
|
|
Fig. 1. Cumulative probability of HCC. |
|
|
Fig. 2. Cumulative probability of decompensation unrelated to HCC. |
|
|
Fig. 3. Cumulative probability of survival. |
Prognostic Factors for HCC Occurrence
In the univariate analysis, 3 baseline parameters were significantly linked to the probability of HCC: history of transfusion, alkaline phosphatase activity above 87 IU/L, and a prothrombin time of 63% or less of the reference value (table 2). The cumulative probability of HCC at 2 and 4 years was significantly lower in IFN-treated than in untreated patients (1.8% and 4.4% vs. 5% and 23%, respectively, P < .001). In multivariate analysis, absence of IFN therapy was the only independent factor predictive of HCC (table 2).
| table 2. Univariate and Multivariate Analyses of Prognostic Factors for HCC Occurrence |
Prognostic Factors for Decompensation Unrelated to HCC
In the univariate analysis, 6 baseline parameters were significantly linked to the probability of decompensation unrelated to HCC: age at diagnosis above 63 years, ALT activity above 76 IU/L, an albumin level below 3.4 g/dL, a prothrombin time of 63% or less of the reference value, a platelet count below 102 × 109/L and a Child-Pugh score B (table 3). The cumulative probability of decompensation unrelated to HCC at 2 and 4 years was significantly lower in IFN-treated than in untreated patients (3.4% and 11% vs. 33% and 38%, respectively, P = .001). In multivariate analysis, absence of IFN therapy was the only independent factor predictive of decompensation unrelated to HCC (table 3).
| table 3. Univariate and Multivariate Analyses of Prognostic Factors for Decompensation Unrelated to HCC |
Prognostic Factors for Survival
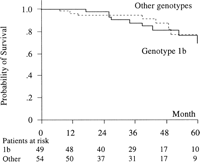
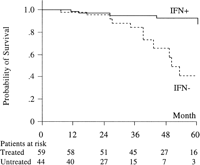
In the univariate analysis, 7 baseline parameters were significantly linked to death or liver transplantation: splenomegaly on physical examination, alkaline phosphatase activity above 87 IU/L, an albumin level below 3.4 g/dL, a prothrombin time of 63% or less than the reference value, a bilirubin level above 1.11 mg/dL, a platelet count below 102 × 109/L, and a Child-Pugh score B (table 4). The probability of survival was not significantly different between patients infected by genotype 1b and patients infected by other genotypes (Fig. 4). Survival at 2 and 4 years was significantly higher in IFN-treated than in untreated patients (97% and 92% vs. 95% and 63% respectively, P < .0001) (Fig. 5). In multivariate analysis, an albumin level below 3.4 g/dL and absence of IFN therapy were the two independent factors predictive of death or liver transplantation (table 4).
| View This table | table 4. Univariate and Multivariate Analyses of Prognostic Factors for Survival |
|
|
Fig. 4. Cumulative probability of survival among patients infected with Genotype 1b (solid line) and patients infected with other genotypes (dashed line). |
|
|
Fig. 5. Cumulative probability of survival among interferon-treated patients (solid line) and untreated patients (dashed line). |
DISCUSSION
This study shows that a large proportion of patients with compensated HCV-related cirrhosis develop complications, regardless of the viral genotype. Independent of other prognostic factors, IFN therapy was associated with significantly lower risk of HCC or decompensation and longer survival.
The patients were homogeneous in terms of recruitment criteria, i.e., primary referred for anti-HCV seropositivity and elevated ALT activity, stage of liver disease at entry (no clinical evidence of decompensation), and follow-up (all patients being evaluated in our department). Only 3% of patients were lost to follow up. The baseline characteristics of patients at entry, mean age 56 years, a majority of men, infected by blood transfusion and harboring genotype 1b, were similar to those in other studies conducted in Western countries. 8,11,12
The cumulative probability of HCC was 3% and 11.5% at 2 and 4 years, respectively, with a yearly incidence of 3.3%, confirming previous studies of Western patients with alcoholic or viral cirrhosis. 8,11 The probability of decompensation which was not related to HCC was quite high, 15% and 20% at 2 and 4 years, respectively. The probability of terminal illness (death or liver transplantation) 2 and 4 years after the diagnosis of cirrhosis was 6% and 16%, respectively. All but one death were related to liver disease. Tong et al. reported a higher prevalence of complications and death among patients with transfusion-associated Hepatitis C; however, the study population was heterogeneous, several patients having HCC or decompensated cirrhosis at the diagnosis of Hepatitis C.7 In a long-term follow-up study, although there was no increased mortality after acute transfusion-associated non-A, non-B Hepatitis, there was a significant increase in the number of deaths related to liver disease.2
We defined prognostic factors for three different endpoints: HCC occurrence, decompensation unrelated to HCC, and death or liver transplantation. As expected, among baseline characteristics, univariate analysis showed that signs of poor liver function and/or portal hypertension were prognostic factors for the three endpoints. Interestingly, Child-Pugh score was found to be a prognostic factor for survival in univariate analyses but not in multivariate analyses, in contrast with albumin level which was a prognostic factor in both analyses. The superiority of albumin level could be caused by the narrow range of Child-Pugh score as only patients with no clinical evidence of decompensation, i.e., without ascites, encephalopathy, or jaundice, were included. For the three endpoints, univariate analyses failed to find any prognostic value of HCV genotype. Several cross-sectional studies have suggested that infection by genotype 1b is associated with more severe liver disease in patients with chronic Hepatitis C,12-14 but this is probably the result of confounding factors such as disease duration.23
A major finding of this study is the role of IFN therapy on outcome of our patients. Indeed, the cumulative probabilities of HCC, decompensation, and death or liver transplantation were significantly lower in IFN-treated than in untreated patients. For each of the three endpoints, the association between IFN therapy and outcome was confirmed in multivariate analyses. The impact of IFN therapy was independent of the biochemical and virological response, only 5% of treated patients having a sustained normalization of transaminases activity associated to HCV RNA PCR negativity. The baseline characteristics of the 44% of patients who were not treated with IFN were not significantly different from those of the treated patients. Before 1992, the majority of our patients with chronic Hepatitis C were treated with IFN; after 1992, most patients with HCV cirrhosis diagnosed in our department were no longer treated with IFN, as several studies, as well as our experience, revealed a very low rate of biochemical or virological response in this population.24-26
The effectiveness of IFN therapy in the prevention of complications of HCV-related cirrhosis is controversial. In a randomized trial, Nishiguchi et al. showed that a 3- to 6-month course of IFN could prevent HCC in patients with HCV-related cirrhosis, independent of the biochemical response.16 The rate of HCC at 4 years was 4% in the treated group compared to 25% in the control group. The study of Mazzella et al. provides similar event although the incidence of HCC was lower and the follow up was shorter.17 In our study, the probability of HCC at 4 years was 4.4% in treated patients and 23% in untreated patients. The mean treatment period (11 months) was longer than in Nishiguchi’s (4 month) study. Survival tended to increase with the IFN therapy duration, without reaching statistical significance (data not shown). This is likely caused by the small number of events in IFN-treated patients. In a recent retrospective study by Fattovich et al. aimed to assess the long-term outcome of compensated HCV-related cirrhosis, univariate analysis showed a significantly higher probability of survival in IFN-treated patients than in untreated patients.27 However, the link between survival and IFN therapy was not confirmed in multivariate analysis. This could be caused by the low yearly incidence (1.4%) of HCC in this cohort as well as by major differences between treated and untreated patients. Indeed, in contrast to our study, the treated patients had the least severe liver disease and, as a result, the prognostic value of liver function was so prominent (e.g., bilirubinemia P < .0001 in multivariate analysis) that the potential effect of IFN may have been masked.
The preventive effect of IFN on decompensation and HCC could be related to properties other than anti-viral action. Indeed, in vitro and in vivo studies have shown an anti-fibrogenic effect of IFN.28-32 Natural-killer cell activity, which is significantly diminished before the onset of HCC in patients with viral cirrhosis,33 is increased by IFN.34 Finally, IFN has also anti-angiogenic properties. 35,36
In conclusion, this retrospective study shows that HCC and decompensation occur in a large proportion of patients with compensated HCV-related cirrhosis. IFN seems to prevent or delay these complications, independent of its anti-viral properties. A prospective controlled randomized trial is needed to confirm these results.
Footnotes
Abbreviations: HCV, Hepatitis C virus; IFN, interferon; HCC, hepatocellular carcinoma; PCR, polymerase chain reaction; ALT, alanine aminotransferase.
Presented in part at the 47th Annual Meeting of the American Association for the Study of Liver Disease, Chicago, IL
Received October 13, 1997; accepted February 13, 1998.
Address reprint requests to: Lawrence Serfaty, M.D., Service d’Hépato-gastroentérologie, Hôpital Saint-Antoine, 184 rue du Faubourg Saint-Antoine, 75571 Paris Cédex 12, France. Fax: 33-1-49-28-21-07.
Copyright © 1998 by the American Association for the Study of Liver Diseases.